
TECHNICAL SPECIFICATIONS
- Working pressure ~10-11mbar
- Si(C-BN-1400 °C), Ge(BN-1200 °C, Mn(BN-1200 °C) K-cells from RIBER; Si (flux=0.04 Å/min); Ge (flux=0.16 Å/min);
- Sb, As, Bi- Surfactants effusion cells;
- Ag, Au- Capping Layer effusion cells;
- DC direct sample heating (RT-1200 °C) and Indirect heating (RT-450 °C ) systems;
- Air-vacuum Fast Load-lock Sample Transfer System;
AVAILABLE TECHNIQUES
-
STAIB EK-3315-R RHEED Ultra-High Vacuum (UHV) System for Surface Science Investigations;
-
Cleaning Semiconductor (SC), Metal (M)-Surfaces reconstruction;
-
Epitaxial growth SC/SC, SC/Metal/SC;
-
Homo- and Hetero-structures growth: 1D, 2D and 3D Materials.
SAMPLES
-
Sample lateral dimensions: 10 x 5 mm (ideal), 3 x 3 mm (minimal), 10 x 10 mm (maximal);
-
Sample thickness: ideally up to 2 mm (thicker and/or smaller samples also feasible).
USED FOR
-
Fundamental Surface Science study;
-
Artificial Atomic Epitaxial Growth;
-
Discovery of new 1D, 2D and 3D epitaxial SC/SC; M/SC for micro-nanoelectronics and solar cells purposes;
-
Semiconductors for Microelectronics;
-
Microcircuits;
-
Ultra-thin Films;
-
Samples Cleaning;
-
Thin-film Stability;
-
Barrier Layers;
-
Lubrication;
-
Chemical Industry;
-
Coatings/Catalysis.
CASE STUDIES
Cross-sectional HRTEM Mn0.06Ge0.94on Ge(001)2x1
The structural, electronic, and magnetic properties of the Mn0.06Ge0.94 diluted magnetic semiconductor, grown at 520 K by molecular-beam epitaxy on Ge(001)2✕1, have been investigated. Diluted and highly ordered alloys, containing Mn5Ge3nanocrystals, were grown. The valence band photoelectron spectrum of Mn0.06Ge0.94 shows a feature located at −4.2 eV below the Fermi level, which is the fingerprint of substitutional Mn atoms in the Ge matrix. Magnetization measurements show the presence of a paramagnetic component due to substitutional Mn atoms and of a ferromagnetic like component due to Mn5Ge3nanocrystallites. The Mn L2,3 x-ray absorption spectrum of this polyphase film shows no marked multiplet structure, but a bandlike character.
See: P. De Padova, et al., Phys. Rev. B 77, 045203 (2008).

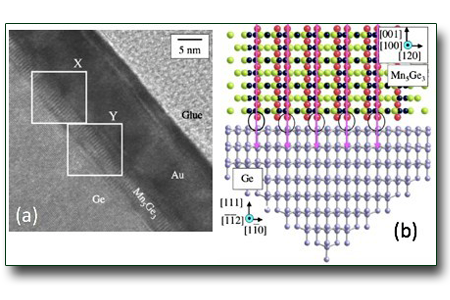
Mn5Ge3 film on Ge(111)
An investigation of the structural, magnetic and electronic properties of≈3 nm thick Mn5Ge3 films epitaxially grown on a Ge(111)-c(2✕8) reconstructed surface is reported. High resolution transmission electron microscopy and selected area electron diffraction give evidence of 2.2% in-plane compressive strain between the Mn5Ge3 film and the Ge substrate. Magneto optical Kerr effect measurements show that the films are ferromagnetic with a Curie temperature of ≈325 K. The analysis of Ge 3d core level photoelectron spectra of the Mn5Ge3 films allows determining an upper limit of 76 meV for the Ge 3d5/2 core-hole lifetime broadening. The Ge 3d3/2 core-hole lifetime broadening is found to be 15 meV larger than that of the Ge 3d5/2 core hole, because of the existence of a Coster–Kronig decay channel due to the metallic character of Mn5Ge3.
See: P. De Padova, et al., Phys. Rev. B 77, 045203 (2008).

 English (UK)
English (UK)  Italiano (Italia)
Italiano (Italia)